- Description
- Reviews (0)
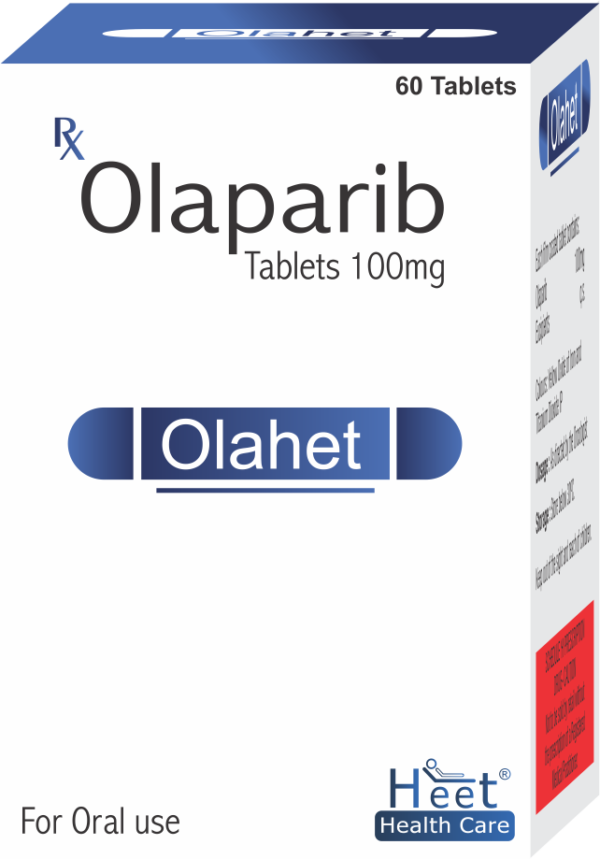
Description
Olaparib 150mg
COMPOSITION
Each film coated contains:
Olaparib…………150MG
Excipients……….Q.S
Colours: Yellow Oxide of iron and Titanium Dioxide I.P.
USE:
Olaparib is a poly (ADP-ribose) polymerase (PARP) inhibitor used to treat ovarian cancer, breast cancer, pancreatic cancer, and prostate cancer.
Indication
Ovarian cancer:
Olaparib is indicated for the maintenance treatment of adults with deleterious or suspected deleterious germline or somatic BRCA-mutated advanced epithelial ovarian, fallopian tube or primary peritoneal cancer who are in complete or partial response to first-line platinum-based chemotherapy.
Olaparib is indicated in combination with bevacizumab for the maintenance treatment of adults with advanced epithelial ovarian, fallopian tube or primary peritoneal cancer who are in complete or partial response to first-line platinum-based chemotherapy and whose cancer is associated with homologous recombination deficiency (HRD)-positive status defined by either: a deleterious or suspected deleterious BRCA mutation, and/or genomic instability.
Olaparib is indicated for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, who are in complete or partial response to platinum-based chemotherapy.
Breast cancer:
Olaparib is indicated for the adjuvant treatment of adult patients with deleterious or suspected deleterious gBRCAm human epidermal growth factor receptor 2 (HER2)-negative high risk early breast cancer who have been treated with neoadjuvant or adjuvant chemotherapy.
Olaparib is indicated for the treatment of adult patients with deleterious or suspected deleterious gBRCAm, HER2-negative metastatic breast cancer, who have been treated with chemotherapy in the neoadjuvant, adjuvant, or metastatic setting. Patients with hormone receptor (HR) positive breast cancer should have been treated with a prior endocrine therapy or be considered inappropriate for endocrine therapy.
Pancreatic cancer:
Olaparib is indicated for the maintenance treatment of adult patients with deleterious or suspected deleterious gBRCAm metastatic pancreatic adenocarcinoma whose disease has not progressed on at least 16 weeks of a first-line platinum-based chemotherapy regimen.
Pharmacodynamics
Olaparib is a cytotoxic and anti-tumour agent. Olaparib inhibits the growth of selective tumour cell lines in vitro and decreases tumour growth in mouse xenograft models of human cancer, both as monotherapy or following platinum-based chemotherapy. The drug exerts anti-tumour effects in cell lines and mouse tumour models with deficiencies in BRCA1/2, ATM, or other genes involved in the homologous recombination repair (HRR) of DNA damage and correlated with platinum response.8
In preclinical models of cancer, olaparib demonstrated anti-tumour activity when used alone, in combination with chemotherapeutic agents, or radiotherapy.5 Olaparib can act as a chemosensitizer to potentiate the cytotoxicity of DNA-damaging chemotherapeutic agents such as alkylating agents and platinum-based drugs. It can also act as a radiosensitizer by preventing PARP-mediated DNA repair.2,4
Mechanism of action
Poly (ADP-ribose) polymerases (PARPs) are multifunctional enzymes comprising 17 members. They are involved in essential cellular functions, such as DNA transcription and DNA repair.8 PARPs recognize and repair cellular DNA damage, such as single-strand breaks (SSBs) and double-strand breaks (DSBs). Different DNA repair pathways exist to repair these DNA damages, including the base excision repair (BER) pathway for SSBs and BRCA-dependent homologous recombination for DSBs.2
Olaparib is a PARP inhibitor: while it acts on PARP1, PARP2, and PARP3, olaparib is a more selective competitive inhibitor of NAD+ at the catalytic site of PARP1 and PARP2. Inhibition of the BER pathway by olaparib leads to the accumulation of unrepaired SSBs, which leads to the formation of DSBs, which is the most toxic form of DNA damage. While BRCA-dependent homologous recombination can repair DSBs in normal cells, this repair pathway is defective in cells with BRCA1/2 mutations, such as certain tumour cells.4 Inhibition of PARP in cancer cells with BRCA mutations leads to genomic instability and apoptotic cell death. This end result is also referred to as synthetic lethality, a phenomenon where the combination of two defects – inhibition of PARP activity and loss of DSB repair by HR – that are otherwise benign when alone, lead to detrimental results.
In vitro studies have shown that olaparib-induced cytotoxicity may involve inhibition of PARP enzymatic activity and increased formation of PARP-DNA complexes, resulting in DNA damage and cancer cell death.
Metabolism
Olaparib is metabolized by cytochrome P450 (CYP) 3A4/5 in vitro. Following an oral dose of radiolabeled olaparib to female patients, unchanged olaparib accounted for 70% of the circulating radioactivity in plasma. Olaparib undergoes oxidation reactions as well as subsequent glucuronide or sulfate conjugation.8 In humans, olaparib can also undergo hydrolysis, hydroxylation, and dehydrogenation.3
While up to 37 metabolites of olaparib were detected in plasma, urine, and feces, the majority of metabolites represent less than 1% of the total administered dose and they have not been fully characterized. The major circulating metabolites are a ring-opened piperazin-3-ol moiety and two mono-oxygenated metabolites. The pharmacodynamic activity of the metabolites is unknown.
Route of elimination:
Following a single dose of radiolabeled olaparib, 86% of the dosed radioactivity was recovered within a seven-day collection period, mostly in the form of metabolites. About 44% of the drug was excreted via the urine and 42% of the dose was excreted via the feces. Following an oral dose of radiolabeled olaparib to female patients, the unchanged drug accounted for 15% and 6% of the radioactivity in urine and feces, respectively.
DOSAGE: As Directed by the oncologist.
STORAGE:
Store



 Your Product is verified
Your Product is verified






Reviews
There are no reviews yet.